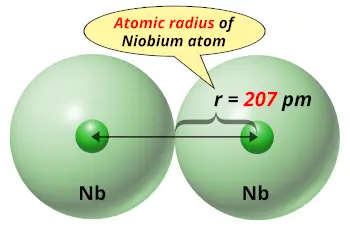 
Most acids therefore do not attack it at room temperature. 
The chemical behavior of niobium is almost identical to that of tantalum, which is directly below niobium in the periodic table.Īs a result of the formation of a passive layer (protective layer), niobium is very resistant to air. As with vanadium, which is above niobium in the periodic table, the +5 level is the most consistent. Niobium is a ductile heavy metal with a gray sheen. If halides are used as the starting material for the reduction, this is done with sodium as the reducing agent. 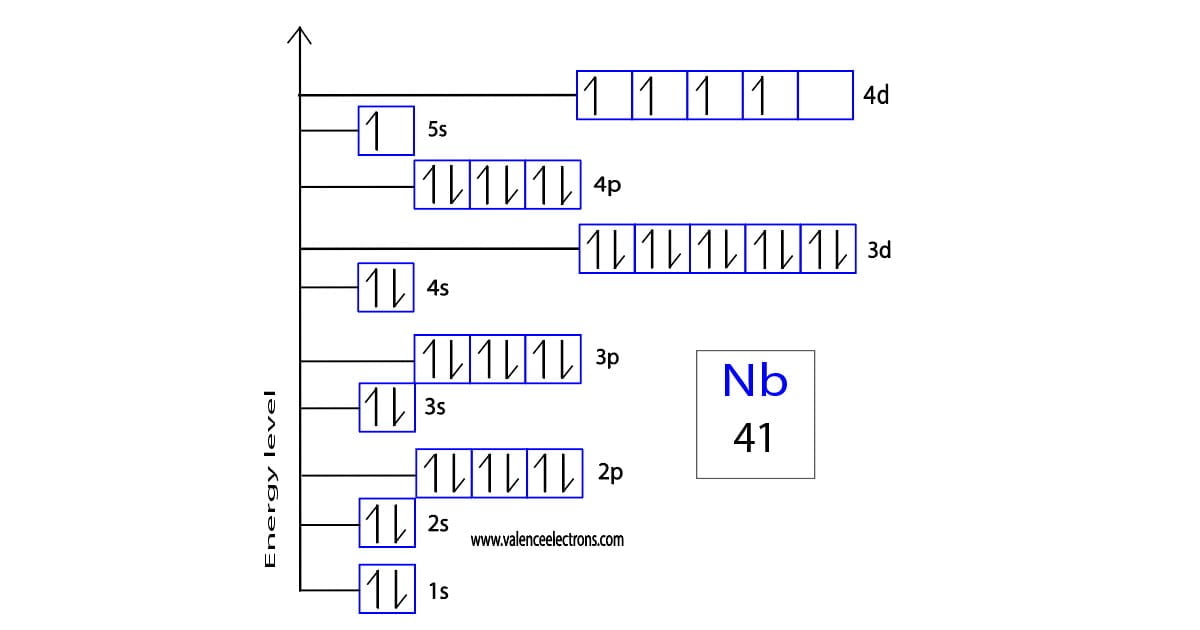
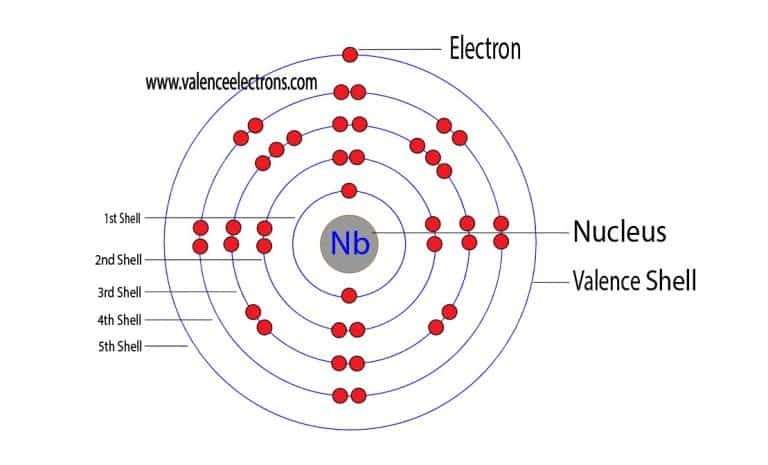
Most of the niobium for the steel industry is produced in this way, with iron oxide being added to obtain an iron-niobium alloy (60% niobium). This is either first converted to niobium carbide with carbon and then reduced to the metal with further niobium pentoxide at 2000 ° C in a vacuum or obtained directly by aluminothermically. Niobium pentoxide is first produced from the separated niobium fluoride by reacting with oxygen. These can be produced by reacting ores, coke and chlorine at high temperatures. A third possibility of separation is by fractional distillation of the chlorides NbCl 5 and TaCl 5. Nowadays, however, separation by extraction with methyl isobutyl ketone is common. The easily soluble niobium fluoride can thus be separated from the tantalum. Only the tantalum fluoride is sparingly soluble in water and precipitates. The dipotassium salts of these fluorides can be formed by converting them into an aqueous phase and adding potassium fluoride. The complex fluorides 2− and 2−that are easily soluble. The first such industrial separation process was developed by Galissard de Marignac in 1866.įirst, the ores are exposed to a mixture of concentrated sulfuric and hydrofluoric acid at 50–80 ° C. Since niobium and tantalum always occur together, niobium and tantalum ores are first digested together and then separated by fractional crystallization or different solubility in organic solvents. Large ore deposits are also located in Nigeria, the Democratic Republic of the Congo, and Russia. Brazil and Canada are the main producers of niobium-containing mineral concentrates. In recent years production has increased significantly. The annual production in 2006 was almost 60.000 t, 90% of which was mined in Brazil. Niobium deposits in carbonatites, where pyrochlore has accumulated in the weathered soils, are of economic interest. The most important minerals are columbite (Fe, Mn) (Nb, Ta) 2O 6, which is also known as niobite or tantalite depending on the content of niobium or tantalum, as well as pyrochlore (NaCaNb 2O 6F). Due to the similar ionic radii, niobium and tantalum are always siblings. Niobium is a rare element with a share of 1,8 In 1907 Werner von Bolton produced very pure niobium by reducing a heptafluoroniobate with sodium. In 1866, Charles Marignac confirmed tantalum as a separate element. In 1864 Christian Wilhelm Blomstrand succeeded in producing metallic niobium by reducing niobium chloride with hydrogen in the heat. It was not until after 100 years of debate that the International Union of Pure and Applied Chemistry (IUPAC) put forward in 1950 niobium as the official name of the element. Not knowing about Hatchett's work and its naming, he named the rediscovered element because of its similarity to tantalum after Niobe, the daughter of Tantalus. It was not until 1844 that the Berlin professor Heinrich Rose showed that niobium and tantalic acid are different substances. Until the middle of the 19th century it was assumed that columbium and tantalum, discovered in 1802, were the same element, since they almost always occur together in minerals (paragenesis). He found it in columbite ore (first find in a river bed in Colombia), which had been sent to England by John Winthrop around 1700. Niobium was discovered by Charles Hatchett in 1801. It is mainly used in metallurgy to make special steels and improve weldability. Niobium can be extracted from the minerals columbite, coltan (columbite-tantalite) and loparite. The rarely occurring heavy metal is gray in color and easily malleable. In the Anglo-Saxon language area is still today many metallurgists, material suppliers and in private use the long outdated designation columbium and the abbreviation Cb . It is one of the transition metals, in the periodic table it is in the 5th period and the 5th subgroup (group 5) or vanadium group. Niobium (after Niobe, the daughter of tantalum) is a chemical element with the element symbol Nb and the atomic number 41. Niobium price, occurrence, extraction and use
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
